BIA 10-2474 THERAPEUTIC TEST GOES FATAL IN FRANCE. WHAT DID HAPPEN ? WHAT ARE THE REAL RISKS IN PHASE I CLINICAL TRIALS ?
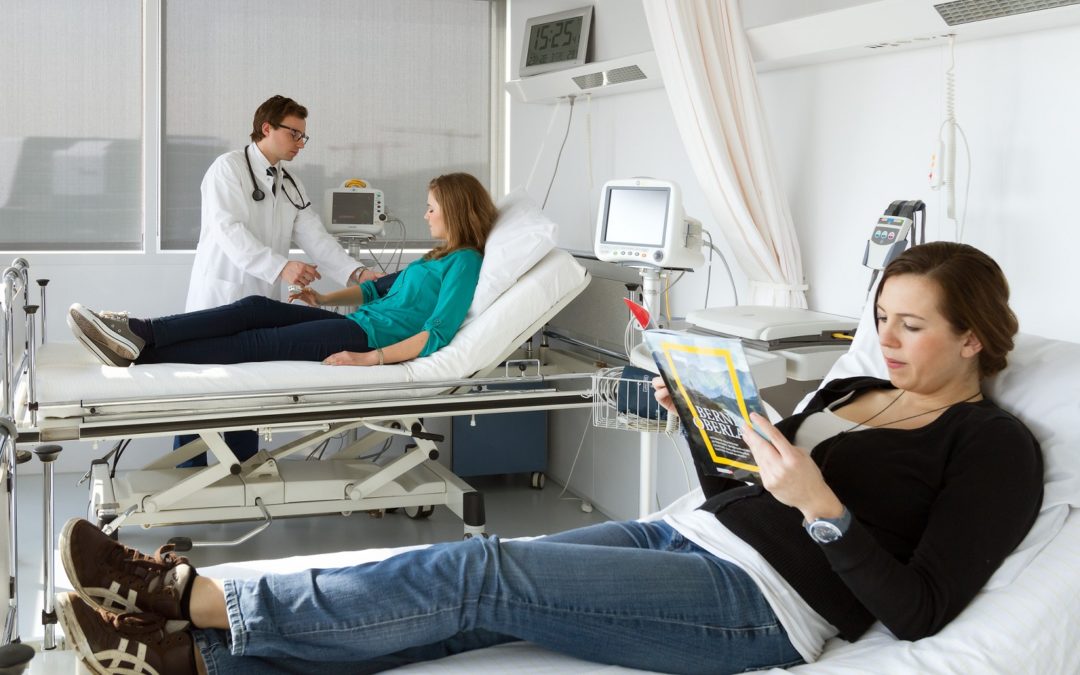
WHAT IS A CLINICAL TRIAL?
A clinical test normally takes place in 3 phases and the drug development may take in total between 10 and 15 years of research between the development of a molecule by a laboratory and the commercialization phase of the study. Phase I (First-In-Men), made after the pre-clinical studies on animals are promising, entails a small group of voluntaries and focuses only in safety. Phase II and III are progressively larger trials on patients with the target disease for assessment of the drug’s effectiveness, although safety remains paramount.
Clinical trials procedures are standardized by the international regulations and are largely the same throughout the world. However, like any safeguard measures, they only minimize risks rather than abolishing them. There is an inherent risk in exposing people to any new compound because the animal model is not fully predictive for the man.
HOW ARE VOLUNTEERS PROTECTED
Studies start at low doses, for a short time and if no unpleasant side effects are reported, continue with another group of volunteers at a higher dose, until they start reporting some adverse reactions like skin rash, nausea, vertigo or other non-critical symptoms. Depending on what is at stake, stronger adverse reactions may be accepted, like hair loss and severe vomiting in cancer treatments
Due to the tests done on animals in the pre-clinical studies, mishaps are relatively uncommon in the clinical phase of the test (in average, once in 26,5 years), but in 2006 after taking part in a Phase I clinical trial on the humanized monoclonal antibody TGN1412 developed as immuno-modulatory drug to fight autoimmune disease and leukemia, six men were treated in London for severe organ failure. Despite being administered at a supposed sub-clinical dose, some 500 times lower than the dose found safe in animals, they developed a strong auto-immune reaction, not observed to similar extent in animals.
WHAT HAPPENED IN FRANCE
In the past few days we learned about a clinical trial that went terribly wrong in France leaving several volunteers in a very serious condition and one dead. Full details are not released so far but it seems that it involved a compound from the Portuguese company Bial and the compound is believed to be BIA 10-2474. The trial in France was led by the contract research organization Biotrial, a contract research organization specialized in testing drugs “between the 4-th and 6-th year of development”, i.e. when the tests pass from animals to humans.
This was a phase 1 clinical trial (First-in-Humans), where healthy volunteers take the molecule BIA 10-2474 developed by Bial, to evaluate the safety of its use, its tolerance and pharmacological profile. The molecule was intended to treat chronic pain and mood disorders such as anxiety, and to improve movement coordination linked to neurodegenerative illnesses. The molecule had never been tested in humans.
The drug candidate was a fatty acid amide hydrolase inhibitor (FAAH). It is an enzyme that degrades endocannabinoid neurotransmitters like anandamide, which relieves pain and can affect eating and sleep patterns. According to the company, since the beginning the protocol has been developed with “respect for international best practice, and with the pre-clinical trials carried out, notably with respect to toxicology” and the trial was approved by the French Regulatory Authority (ANSM) and the Brest Regional Ethics Committee.
In a statement to the media on 15 January the French Health Minister Marisol Touraine stated that it had been previously validated in animals, including chimpanzees, as provided in this highly controlled process. The test on chimpanzees seems to has been finished the same month when the human study started, which is not unusual practice. Animal toxicology studies need to be of the same duration as the planned clinical trial and Phase I trials are of short duration. So far few details of the pre-clinical testing of this molecule have been made public and it is not clear how long the animal test was performed.
According to the protocol, the trial enrolled non-smoking men and women aged between 18 and 55 who had a body mass index between 19 and 30. The plan revealed by a rejected study candidate was to give patients BIA 10-2474 every day between the third and thirteenth day of their stay at the clinic, while they underwent an extensive battery of tests and sampling–including as many as nine blood collections on some days. On the first and last day of drug administration, patients’ heart rates were to be monitored around the clock while all of their urine was collected for analysis. (Between day 3 and 9, however, all they apparently had to do was take BIA 10-2474 and provide a single blood sample). They were due to be released on 18 January but had to come back for a final check-up and more sampling on 1 February.
It seems however that part of the last group of 6, which was receiving the highest dose, have been out-patients and not monitored in the hospital of Rennes, probably due to the lack of moderate or severe adverse effects so far. Eg. they were not in the company settings when the adverse reactions started because they were admitted in different hospitals. The company setting also doesn’t seem to have an intensive care, as they were using the ICU of the hospital of Rennes, which is in proximity. This doesn’t seem to be the best practice for Phase I studies, especially in high risk studies with little experience from similar compounds having been tested.
The study employed a 3-stage design with 108 volunteers in good health already participated in the testing of this new molecule at lower doses without developing adverse reaction, neither serious, nor moderate. French Health Minister Marisol Touraine said that to date, 90 of them were already given variable doses of the test drug candidate with no reported ill effects, the others – placebo. Phase I studies don’t usually have a placebo group; it’s not clear why this one did. The trial would first test a single dose, then multiple doses, and finally the combination of the drug with a meal. Some volunteers received the molecule in a single dose, other multiples and this at different doses.
In the third stage of the trial evaluating multiple doses, six male volunteers received doses orally. They had repeatedly taken the medication. Four of them are suffering injuries including deep hemorrhagic and necrotic lesions in the brain. The first volunteer was hospitalized at the Rennes University Hospital on January 10, became brain dead and died on January 17. Bial says that the new drug had already been administered to 108 patients “without any moderate or serious adverse reaction.” However it is not clear what mild side effects have been seen and has the protocol been proactively updated to take them in consideration. Besides phase I clinical trials usually start with single or lower doses; the six patients may have been the first to receive multiple doses.
The six people hospitalized have all received “the highest dose, repeated doses,” recounts Pierre-Gilles Edan, head of neuroscience at the University Hospital of Rennes center. The first shot was administered on Thursday, January 7th. The first symptoms appeared on Sunday, January 10 for the first patient, currently brain dead. The volunteer was admitted to in intensive care of the hospital in Rennes on Monday. This patient was hospitalized in a state that was deteriorating rapidly, leaving initially considering a stroke.
The other five were gradually hospitalized on Wednesday and Thursday during the week. “Today four have severe neurologic disorders, which makes us fear, in the best of situations, an irreversible disability,” said Pierre-Gilles Edan. The fifth has for the moment no symptoms, but is constantly monitored. The doctors are trying to control the inflammatory reaction with corticosteroids but no antidote to this drug is known today.
It is not yet clear why many others who participated in the study since it began in July apparently haven’t experienced similarly severe side-effects. It’s possible that BIA 10-2474 drug had completely unexpected effects because it binds to another target besides FAAH, said molecular pharmacologist Stephen Alexander of the University of Nottingham in the U.K. in a statement distributed by the Science Media Center (SMC); such “off-target” interactions might only become dangerous with more extensive exposure. “Another option is a dosing accident, where patients were given far more than clinicians thought were in the dosage form,” added the neuropharmacologist Ben Whalley of the University of Reading , also in an SMC statement. It’s possible that the six were the first to receive multiple doses of the drug, or or that a batch of drug was contaminated in some fashion
Biotrial interrupted the trial on January 11 and all the volunteers who participated earlier have been recalled to undergo brain scans and other testing. The men have now apparently a higher risk of cancer and autoimmune diseases tied to their exposure to the experimental drug.
WHAT WAS THE MOLECULE?
It was a molecule of the laboratory Portuguese Bial. The drug was developed especially to treat movement disorders associated with neurodegenerative diseases. The molecule acts on cannabinoid receptors in the human body, but it is not a drug derived from cannabis. Many other pharmaceutical companies, including Merck, Pfizer, Johnson & Johnson, and Vernalis, have previously tested other FAAH inhibitors into clinical trials without experiencing such adverse events. Related enzyme inhibitor compounds have been sold illicitly as designer drugs, all without reports of this type of toxicity emerging, so the mechanism of the toxicity observed with BIA 10-2474 remains poorly understood.
LESSONS LEARNED FROM THE TGN1412 STUDY
In December 2006, the final report of the Expert Group on Phase One Clinical Trials was published. It found that the trial had not considered what constituted a safe dose in humans, and that the then current law had not required it. The UK National Institute for Biological Standards and Control wrote in 2009 that a near-maximum immuno-stimulatory dose had been given, because a safe starting dose in man had been calculated “based upon results from pre-clinical safety testing in a non-responsive species“.
It made 22 recommendations, including the need for independent expert advice before a high risk study was allowed, testing only one volunteer at a time (sequential inclusion of participants) in case there were rapid ill effects, and to give drugs slowly by infusion instead of an injection. The European guidelines for first-in-man phase-I clinical trials of biologics had been revised.
The predictive value of pre-clinical animal models required reevaluation, dose fixing needed refinement or redesign, criteria for high-risk antibodies needed to be established, and pre-Phase I studies were needed where a dose is calculated with a pre-clinical No-effect level instead of the No-observed-adverse-effect level.
PENDING QUESTIONS
According to the opinion of Judy Stone published in Forbes on January 16, there are many questions remaining open, as little information has been released. We don’t know what doses this group of ill volunteers received, or how that was different from earlier groups. It was likely the first of a higher dosage. We don’t know if or how food affected the drug’s metabolism. Could there have been a contaminant causing disaster in one batch of drug? While much less likely with an oral than IV drug, an error in manufacturing or the concentration of drug could perhaps have affected one batch and not others. Why was there a large placebo group, given that is atypical in Phase 1 trials? Did these patients all receive the drug at the same time (in parallel) or sequentially? It appears that they all received it on January 7. In the last disastrous clinical trial, the TeGenero TGN1412 in London, in 2006, six Phase 1 healthy volunteers almost died with multi-organ failure from a cytokine storm. This became known as the “Elephant Man” trial, for how bad the victims looked. One of the major lessons was that volunteers should be dosed sequentially, allowing time between each to detect a serious adverse reaction. Why didn’t that happen here?
Volunteers safety is paramount for clinical trials to recruit participants and 1/3 of the studies fail to enroll sufficient number of patients to provide statistically meaningful results, especially in cancer studies, where only 20% of the trials reach their recruitment goals. To gain patient confidence, more stringent rules are needed and more transparency. Otherwise the development of new drugs will stall.
STUDY INFORMATION SHEET
Study description
To register, thank you to call for FREE 0800-503-460 (toll free from a landline)
Tested product
BIA 10-2474 is a product under development for the treatment of various medical disorders ranging from anxiety disorders of Parkinson’s disease motor disorders but also in the treatment of chronic pain of multiple sclerosis, cancer has high blood pressure or in the treatment of obesity … The main objective is to evaluate the safety and tolerability of the administration, oral single dose
Allowance: € 1,900 (including travel expenses)
Dates of study: You will need to perform all of the dates selected group.
Date and place of Group Hospitalization selection examination in Rennes End of study
5 Wednesday 09, Thursday 10 and Friday, 11/12/2015 From 04/01/2016 to 18/01/2016 On 01/02/2016
Course of study
1) Selection Examination (medical examination is not compensated).
Ø A visit to the center fasting Biotrial Rennes and Nantes lasting 2 to 3 hours will be necessary: it will aim to take stock of your health and will clarify whether it is compatible with your entry into the ‘test. During the visit, the following tests will be carried out after signing the consent form: a complete physical examination, electrocardiogram, blood pressure measurement and heart rate, blood test and urine, a breath test, pregnancy test for women capacity to procreate.
Ø Documents required obligatorily (we can accommodate you without these documents)
- Your paper certificate of affiliation to the Social Security or equivalent valid, less than a year (not accepted Carte Vitale)
- A piece of identification with a photo or a residence permit for foreign nationals
Ø Instructions to follow before the screening visit:
- No physical activity leading to a muscular effort in the previous 7 days
- 1.5 liter of water per day during the previous 7 days
- No alcohol within 48 hours
(If the results of the various tests are satisfactory, we will confirm your input étudeJ
2) Planning of the study:
The study includes 1 Hospitalization (only at Rennes) and is as follows:
Day -2: Entry to Biotrial to 18H
- urine and blood Samples
- Measurement of vital signs *
- urine pregnancy test for women
- Toxic Search in urine
Day-1:
9 • Blood samples
- Measurement of vital signs *
Day 1:
- Product Administration
- Measurement of vital signs *
9 • Blood samples
- continuous urine on 24 Digest
- Telemetry (painless recording of the activity of the heart) for 24 hours
Day 2:
- Product Administration
- Blood sample
- Measurement of vital signs *
- End record of heart activity
- End of continuous urine collection over 24 hours
Ø Days 3, 4, 5, 6, 7,8 and 9
- Product Administration
- Blood samples
Ø Day 10:
- Product Administration
9 • Blood samples
- Measurement of vital signs *
- A continuous urine collection over 24 hours
- Telemetry (enregistrementindolore the activity of the heart) for 24 hours
Ø 11 Days:
- End record of heart activity
- Measurement of vital signs *
- Blood sample
- A continuous urine collection over 24 hours
Ø 12 Day:
- Measurement of vital signs *
- Blood samples
- A continuous urine collection over 24 hours
Ø 13 Day:
- Measurement of vital signs *
- Blood samples
- End of the morning urine collection
- Urine Analysis
- Released the center after breakfast
Ø end of study visit:
- Complete Physical Examination
- Measurement of vital signs *
- Urine and blood withdrawals, blood pregnancy test for women.
- Temperature, weight, blood pressure, respiratory rate, electrocardiogram and breath test
To participate in this study you must:
- Being a man or a woman aged 18 to 55 years inclusive
- Have a good venous capital
- Non smoking be or have stopped smoking for at least three months
- Have a body mass index between 19 and 30 (BMI)
- Have the results of the clinical examination compatible with the selection criteria
- Have a social cover (paper certificate affiliate to Social Security or equivalent valid and less than a year, the health card is not accepted)
- Not having made a donation of blood, had received a transfusion or blood loss in the three months preceding the study
- Not having made a donation of plasma or platelets in the previous 14 days
- Not having taken medication within 14 days prior to the study
- Have not previously participated in a clinical trial involving the same product
- Do not participate in a trial with or without a therapeutic purpose in the three months preceding the study
- For women of reproductive capacity (Excluded when pregnant or during lactation)
Contraception: either be sterilized surgical or use a non-hormonal contraceptive manner (IUD copper, condom, diaphragm, cervical cap associated with spermicide, or abstinence.
For menopausal women taking hormone replacement therapy: This treatment must be stopped 28 days before the first administration of the product until the end of the study.